- Case-Based Roundtable
- General Dermatology
- Eczema
- Chronic Hand Eczema
- Alopecia
- Aesthetics
- Vitiligo
- COVID-19
- Actinic Keratosis
- Precision Medicine and Biologics
- Rare Disease
- Wound Care
- Rosacea
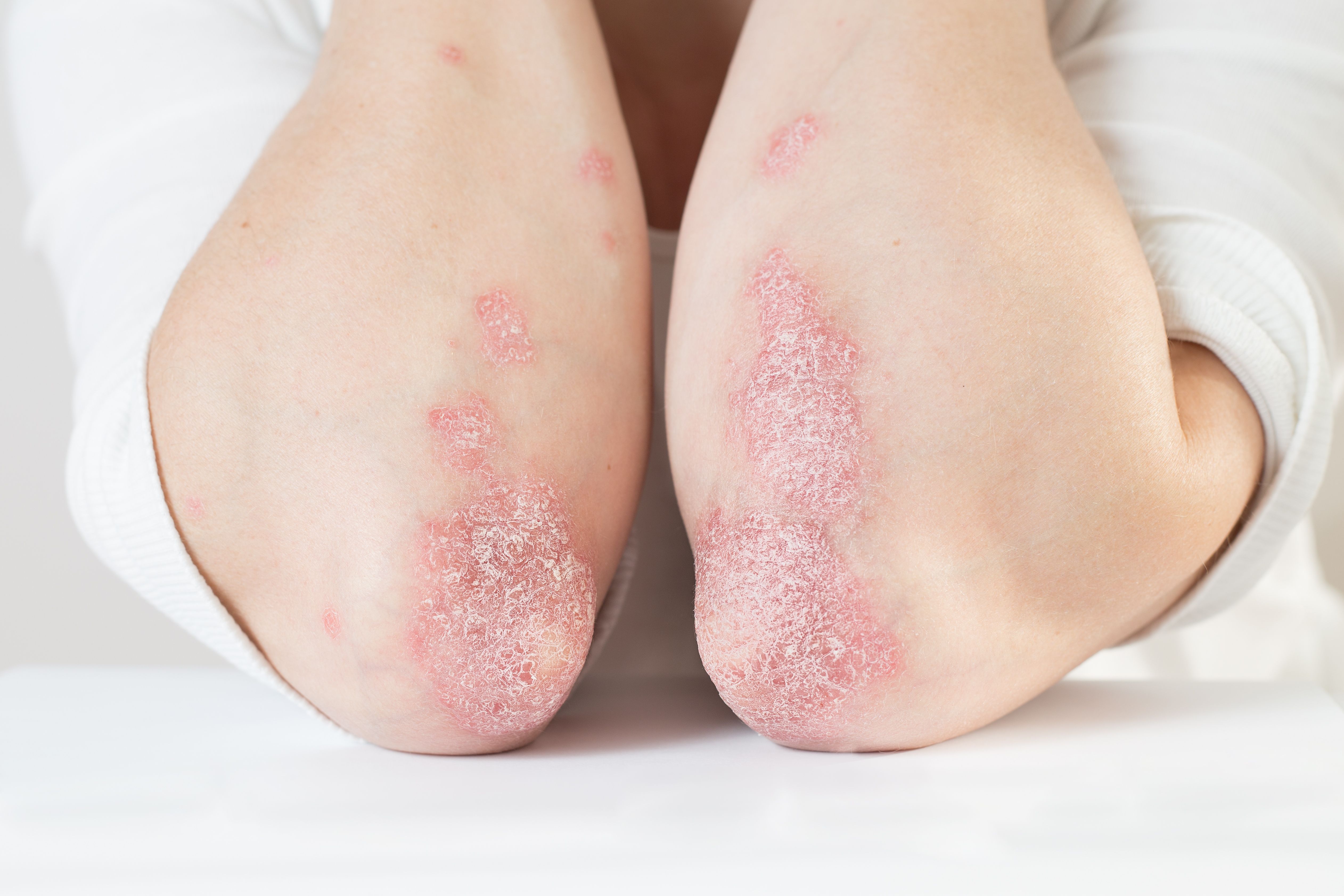
- Psoriasis
- Psoriatic Arthritis
- Atopic Dermatitis
- Melasma
- NP and PA
- Skin Cancer
- Hidradenitis Suppurativa
- Drug Watch
- Pigmentary Disorders
- Acne
- Pediatric Dermatology
- Practice Management
- Prurigo Nodularis
- Buy-and-Bill
Publication
Article
Dermatology Times
IL-23 inhibitors may offer psoriasis patients longer-lasting results
Author(s):
With extended remissions, high safety and favorable dosing schedules, interleukin (IL)-23 inhibitors are well-suited for a variety of patients with psoriasis, says an expert who spoke at the South Beach Symposium
Dr. Lebwohl

With extended remissions, high safety and favorable dosing schedules, interleukin (IL)-23 inhibitors are well-suited for a variety of patients with psoriasis, says an expert who spoke at the South Beach Symposium.
RELATED: Phase 3 results show risankizumab outperforms secukinumab
The IL-23 inhibitors’ mechanism of action and infrequent dosing schedule provide advantages over previous-generation biologics, says Mark Lebwohl, M.D. He is the Waldman Professor and Chairman, Kimberly and Eric J. Waldman Department of Dermatology, Icahn School of Medicine at Mount Sinai.
“In the pathophysiology of psoriasis, the key target appears to be IL-17,” he says.
The IL-23 blockers guselkumab, tildrakizumab, risankizumab and mirikizumab block the cytokine that upregulates the Th17 cell, which makes IL-17. The Th17 cell not only stops making IL-17, but then either hibernates or withers away, according to Dr. Lebwohl.
“Then it takes several months to reconstitute those cells. So these drugs work more slowly than the IL-17 inhibitors, but their effect is longer lasting,” he adds.
Brodalumab, ixekizumab and secukinumab can provide 50% Psoriasis Area and Severity Index (PASI) reductions in 1.8 to 3 weeks, but with IL-23 inhibitors, “patients really only start getting going after four weeks,” says Dr. Lebwohl.
But the IL-23 blockers are highly efficacious, he notes, and results typically peak around week 22. Once patients reach maximum efficacy, he adds, they typically maintain response for years. For example, guselkumab phase 3 data shows patients maintaining PASI 75, 90 and 100 response levels through 156 weeks. Risankizumab has shown similar success through 136 weeks and tildrakizumab through five years, he says.
RELATED: Leg psoriasis improves with topical lotion
Additionally, IL-23 inhibitors can provide extended remission while patients are off therapy. In tildrakizumab trials, the proportion of patients who lost 50% of their PASI response after 48 weeks off therapy was 51%. This figure includes 47% at the 200 mg dose and 54% at 100 mg (the latter dose is approved in Europe but not the United States).
Because it takes so long for psoriasis to return, says Dr. Lebwohl, IL-23 blockers can be given as infrequently as every two or three months. Guselkumab, tildrakizumab and risankizumab are all given at weeks zero and four, then every eight (guselkumab) or 12 (tildrakizumab, risankizumab) weeks thereafter.
These drugs also appear to be extremely safe, he adds.
“Based on the little data we have - mostly from animal studies - it appears that they do not cause an increase in malignancies. We don’t have enough data to say that for sure. But they don’t carry black box warnings about infection or malignancy, which we see in the TNF blockers’ package inserts.”
Considering the above advantages, Dr. Lebwohl says, “when you’re having trouble adequately treating a patient with moderate disease, you wonder if it’s worth a safe injection every three months to keep them clear.”
An analysis of guselkumab phase 3 data examining patients with moderate disease showed that with injections every two months, 71.8% and 77.2% of patients achieved PASI 90 at weeks 16 and 24, respectively, versus 48.2% and 54.1% for adalimumab (p < 0.001).
RELATED: Biologic for psoriasis temporarily impacts aortic vascular inflammation
Altogether, according to Dr. Lebwohl, the IL-23 blockers’ efficacy, dosing schedules and extended remissions make them ideal for patients on Medicare Part D. After these patients reach the “doughnut hole,” they must pay the amount of the doughnut hole plus 5% of their drug costs above the doughnut hole.
“That comes to about $9,000 a year for some drugs,” says Dr. Lebwohl. “Many patients can’t afford that.”
Because IL-23 blockers are administered in hospitals, however, they count toward patients’ medical benefits. “So it doesn’t cost the patient anything,” he notes, “That’s a real plus.”
Additional patients for whom these drugs are proving especially useful include those who travel frequently, or cannot get to the hospital regularly, such as prisoners or those in other institutions.
“They can get injections as infrequently as every three months. And then there are patients who are difficult to inject, such as those with Parkinson’s disease. This is ideal - they only come in four times a year to get the injection.”
RELATED: IL-17 potential therapeutic target for pustular psoriasis
The high efficacy of IL-23 inhibitors means that unlike with most biologic drugs, obese patients lose very little response. The category shows promise for psoriatic arthritis, Dr. Lebwohl adds, although IL-17 inhibitors presently dominate this indication.
Disclosures:
Dr. Lebwohl has been an investigator and advisor for guselkumab (Janssen), tildrakizumab (Sun Pharma), risankizumab (AbbVie) and mirikizumab (Eli Lilly), and most of the biologic therapies available for psoriasis. All payments for research go to his employer, the Icahn School of Medicine at Mount Sinai, and he receives no personal direct compensation from the manufacturers of biologic therapies.
References:
Mark Lebwohl MD. “Tips in the Use of Anti-IL-23 Antibodies,” South Beach Symposium. February 8, 2020.