- General Dermatology
- Eczema
- Chronic Hand Eczema
- Alopecia
- Aesthetics
- Vitiligo
- COVID-19
- Actinic Keratosis
- Precision Medicine and Biologics
- Rare Disease
- Wound Care
- Rosacea
- Psoriasis
- Psoriatic Arthritis
- Atopic Dermatitis
- Melasma
- NP and PA
- Skin Cancer
- Hidradenitis Suppurativa
- Drug Watch
- Pigmentary Disorders
- Acne
- Pediatric Dermatology
- Practice Management
- Prurigo Nodularis
News
Article
FDA Approves Secukinumab for Hidradenitis Suppurativa
Author(s):
Secukinumab is currently the only IL-17A inhibitor approved for the condition.
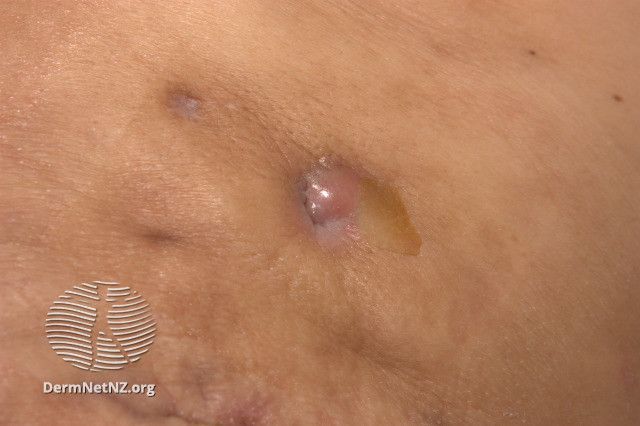
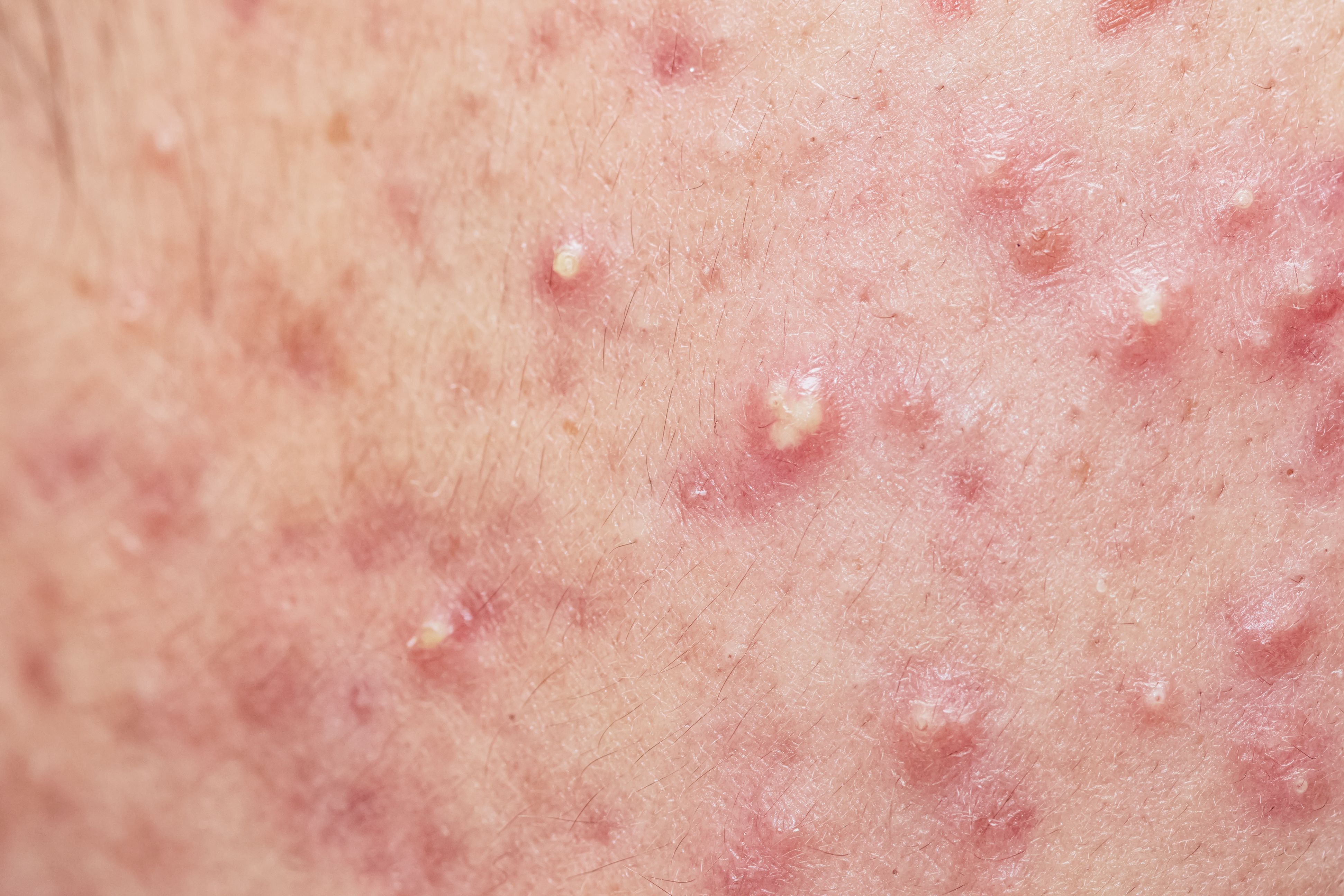
The FDA has approved secukinumab (Cosentyx, Novartis) for the treatment of moderate to severe hidradenitis suppurativa (HS) and is currently the only anti-interleukin (IL)-17A inhibitor approved for the condition. The drug is already approved in the US for the treatment of plaque psoriasis, psoriatic arthiritis, ankylosing spondylitis, and non-radiographic axial spondylarthritis.1
“The FDA has made Halloween special for dermatology, approving Cosentyx for use in hidradenitis suppurativa patients," Christopher Bunick, MD, PhD, associate professor of dermatology and physician-scientist at the Yale School of Medicine in New Haven, Connecticut, told Dermatology Times in response to the approval.
He added, "This is no trick, but rather a real treat for HS patients who often suffer from severe and debilitating skin lesions. HS patients have had very limited advanced systemic therapy options, but today’s FDA approval really gives new hope to HS patients who need biologic therapy. Now in addition to Humira, an anti-TNF alpha biologic, dermatologists have an IL-17A inhibitor available to use for moderate to severe HS. This is a real game changer in the management of HS and will enable more patients to receive potentially life altering therapy."
In an analysis of the SUNSHINE (NCT03713619) and SUNRISE (NCT03713632) phase 3 trials, which evaluated the safety and efficacy of the IL-17A therapy in adult patients with moderate to severe HS, investigators sought to identify the effects of secukinumab on the number of draining tunnels present in patients. The analysis examined its effects in both the general population of patients among the studies and in patients with 1 or more draining tunnel at baseline.2
Read previous Dermatology Times coverage of the SUNSHINE and SUNRISE trials here.
A total of 1084 patients from the SUNSHINE and SUNRISE trials were included in the analysis, including 361 patients who were treated with secukinumab on a biweekly basis, 360 patients who received treatment with secukinumab every 4 weeks, and 363 patients who had been assigned to a placebo.
At baseline, 66.2% of patients receiving secukinumab every 2 weeks presented with at least 1 draining tunnel. 60.6% of patients treated with secukinumab every 4 weeks and 62.5% of patients receiving a placebo treatment presented with 1 or more draining tunnel.
In patients receiving either dosing of secukinumab, the average decrease in number of draining tunnels from baseline to week 16 of the study was greater than in patients receiving a placebo.
Average Draining Tunnels Decrease From Baseline to Week 16
- Secukinumab every 2 weeks: −1.4 ± 2.95
- Secukinumab every 4 weeks: −1.0 ± 2.79
- Placebo: −0.6 ± 2.94
Additionally, a higher proportion of patients treated with either dose of secukinumab did not experience an increase in draining tunnels from baseline to week 52. This was sustained through week 52 of the study.
“These findings suggest that in patients with moderate to severe HS, secukinumab was effective in reducing the number of draining tunnels at week 16, with the effects being sustained through week 52,” study authors wrote. “In addition, at week 52, more than 80% of patients treated with secukinumab did not experience an increase in the number of draining tunnels from baseline, which is relevant because skin tunnel formation is associated with progression of disease in HS and indicates irreversible tissue damage.”
References
1. Novartis. FDA approves Novartis Cosentyx® as the first new biologic treatment option for hidradenitis suppurativa patients in nearly a decade. Press release. Published online October 31, 2023. Accessed October 31, 2023. https://www.novartis.com/us-en/news/media-releases/fda-approves-novartis-cosentyx-first-new-biologic-treatment-option-hidradenitis-suppurativa-patients-nearly-decade.
2. Bechara F, Becherel P-A, Reguiai Z, et al. Effect of secukinumab on draining tunnels in patients with moderate to severe hidradenitis suppurativa: post hoc analysis of the SUNSHINE and SUNRISE phase 3 randomized trials. Poster presented at the 2023 Society of Dermatology Physician Assistants Fall Conference, October 26-29; Nashville, TN.