- Case-Based Roundtable
- General Dermatology
- Eczema
- Chronic Hand Eczema
- Alopecia
- Aesthetics
- Vitiligo
- COVID-19
- Actinic Keratosis
- Precision Medicine and Biologics
- Rare Disease
- Wound Care
- Rosacea
- Psoriasis
- Psoriatic Arthritis
- Atopic Dermatitis
- Melasma
- NP and PA
- Skin Cancer
- Hidradenitis Suppurativa
- Drug Watch
- Pigmentary Disorders
- Acne
- Pediatric Dermatology
- Practice Management
- Prurigo Nodularis
- Buy-and-Bill
Article
Disease burden high for adults with atopic dermatitits
Author(s):
Patients with moderate-severe atopic dermatitis are at high risk of having inadequate disease control, according to a cross-sectional study in JAMA Dermatology.
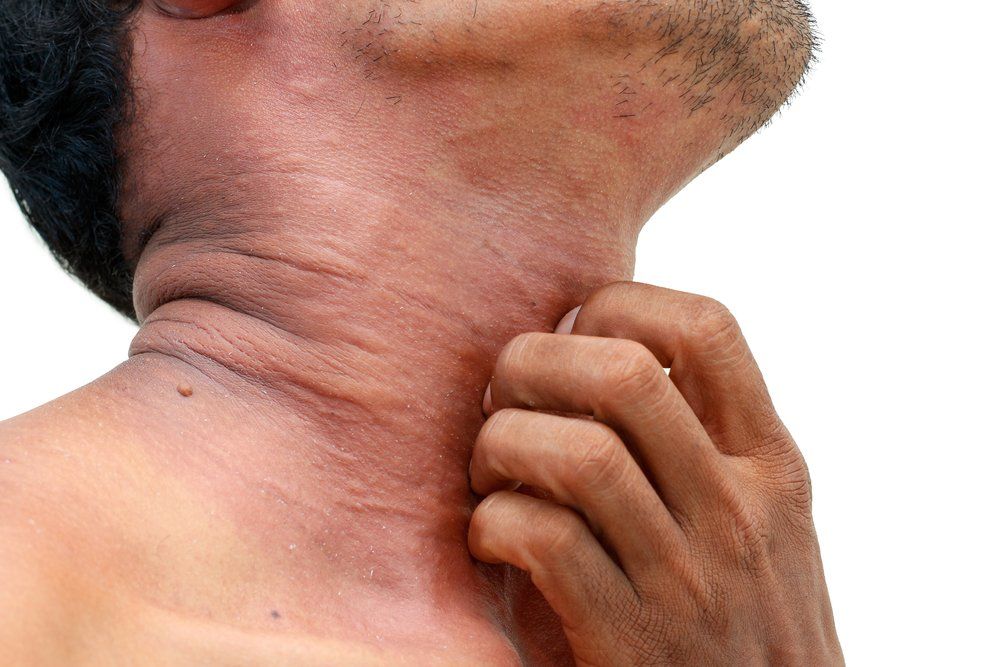
Patients with moderate-severe atopic dermatitis are at high risk of having inadequate disease control, according to a cross-sectional study in JAMA Dermatology.
“It was surprising that despite treatment of moderate-severe disease with our traditional systemic treatment, over half of patients felt like their disease was not adequately controlled,” said Eric Simpson, M.D., of Oregon Health and Sciences University. “This identifies the need for improved systemic therapy for atopic dermatitis, prior to the approval of dupilumab.”
Dr. Simpson was inspired to undertake the study to better understand the burden of adult atopic dermatitis in a population from a clinic rather than a trial population.
The study used data from six academic medical centers in the United States, which collected information from a patient self-administered internet questionnaire.
A total of 1,519 adults patients with atopic dermatitis were stratified by disease severity as mild or moderate-severe using the Patient-Oriented-Scoring Atopic Dermatitis (PO-SCORAD).
Compared to patients with mild atopic dermatitis (n = 689), patients with moderate-severe disease (n = 830) reported more severe itching and pain, greater sleep adverse events, higher prevalence of anxiety and depression, and greater health-related quality-of-life impairment.
Patients with moderate-severe disease who were treated with systemic immunomodulations or phototherapy within the past seven days were further stratified as having adequate or inadequate disease control.
In all, 22.3% (n = 185) of patients with moderate-severe disease were treated with the above treatment protocol, of whom 55.7% (n= 103) considered themselves inadequately controlled.
Patients with inadequately controlled atopic dermatitis were younger than those with controlled disease: 44.6 years vs. 49.8 years.
However, there were no differences between the controlled and inadequately controlled group for age of onset, disease duration or use of topical therapies and any systemic treatments.
The pruritus numerical rating scale (0 to 10, with 0 being no itch and 10 being worst imaginable itch within the past 24 hours) was 2.0 for mild disease, 5.9 for moderate-severe disease, 5.4 for moderate-disease patients treated with immunomodulations or phototherapy who felt their disease was controlled, and 6.9 for inadequately controlled treated patients.
Patients with moderate-severe disease also experienced more days per week with itchy skin compared to mild disease: 5.7 vs. 2.7. Also, itch duration greater than half a day: 22.8% of moderate-severe patients vs. 2.9% of mild patients.
Pain severity was also higher among moderate-severe patients (3.3 on a scale of 0-10, compared to 0.9 for mild disease), and for inadequately controlled vs. controlled disease (3.2 vs. 4.7).
In addition, moderate-severe patients were more likely to have trouble sleeping: 3.9 vs. 1.1, based on the PO-SCORAD visual analog scale (VAS), as well as longer sleep latency (38.8 minutes vs. 21.6 minutes), more frequent sleep disturbances (2.6 nights in the past week vs. 0.4 nights) and greater need for over-the-counter sleep medication (39 percent of patients vs. 21 percent).
Sleep problems rated “much” or “very much” interfered with daily function much more often in patients with moderate-severe atopic dermatitis: 24.3 percent vs. 6.8 percent for mild disease.
Likewise, mental health symptoms of anxiety and depression were reported by 50.2 percent of patients with moderate-severe disease compared to 27.3 percent with mild disease.
The mean Dermatology Life Quality Index (DLQI) scores were also higher in patients with moderate-severe disease (9.2 vs. 2.9), and among patients with inadequately controlled as opposed to controlled disease (13.4 vs. 9.3).
For item 7 on the DLQI, 14.0 percent of moderate-severe patients reported that their disease prevented work or study compared to only 2.6 percent of mild patients. Similarly, the percentages were 27.2 percent and 18.3 percent for inadequately controlled and controlled disease, respectively.
“In contrast to studies that have reported few differences in the burden between mild and moderate/severe AD, the current study reports a significantly greater burden of moderate/severe AD across all outcomes compared with mildly affected patients,” the authors wrote.
These differences may be due to sample size limitations of the other studies or from differences in assessing disease severity.
“Clinicians need to explore more thoroughly how the disease affects their patients on a day-to-day basis,” Dr. Simpson says. “Such exploration will likely lead clinicians to offer more aggressive forms of therapy to help alleviate patient suffering.”
DISCLOSURES
Dr. Simpson is a consultant for Regeneron Pharmaceuticals and Sanofi.
REFERENCE
Simpson EL, Guttman-Yassky E, Margolis DJ, et al. “Association of Inadequately Controlled Disease and Disease Severity With Patient-Reported Disease Burden in Adults With Atopic Dermatitis.” JAMA Dermatol. Published online July 3, 2018. DOI:10.1001/jamadermatol.2018.1572.